[ precision medicine ]
for Autoimmune Disease, start with MS
Autoimmune disease burden
in US and globally
Tempus AI$1.59B rev (2025)
Guardant Health$780M rev (2025)
Exact Sciences$2.8B rev (2025)
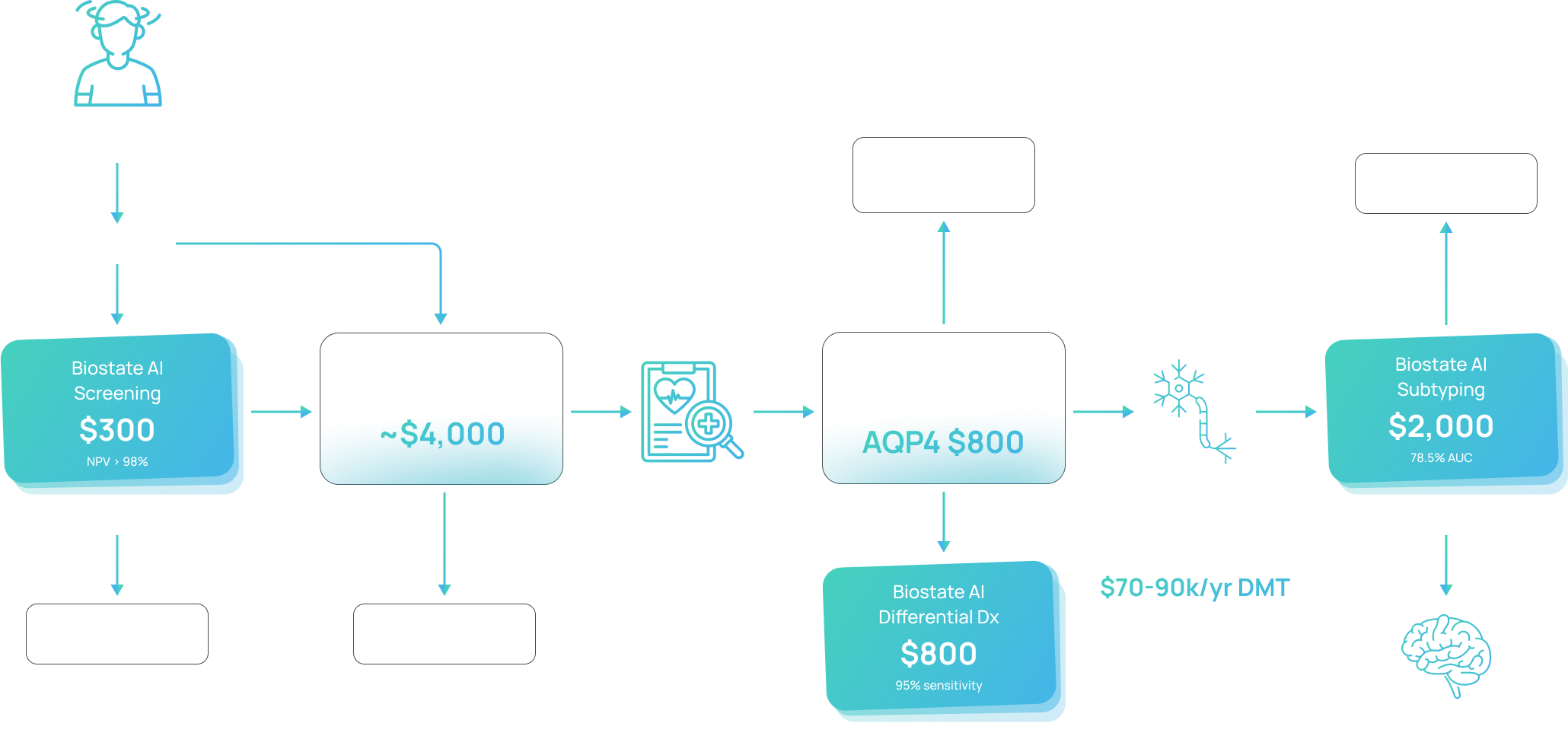
Blood-based assays at three decision points in the MS workflow, replacing or complementing imaging and CSF analysis at a fraction of the cost.
One platform, three markets
Misdiagnosis between MS and NMO can be catastrophic — NMO is worsened by standard MS therapies. BIRT reads the underlying B-cell biology of NMO to catch cases the AQP4-IgG CBA misses (~25% of NMO is seronegative).
The RRMS→SPMS transition is currently invisible. Neurologists wait 2–5 years for retrospective clinical confirmation while irreversible damage accumulates. BIRT provides a prospective molecular signal — annual screening, mammography analog.
Before MRI, a $300 blood draw rules out MS with NPV > 98%. A high-sensitivity screen that avoids unnecessary $4,000 MRIs and shortens the 4–7 year diagnostic delay.
One blood draw, three actionable clinical results
NMO Diagnosis: Wrong Drug = Catastrophic Harm
MS subtyping: detect progression before irreversible damage
@80% cov
/ yr
MS Screening: Blood-First Triage Before MRI
BIRT vs. MRI: Complementary, Not Competing
| 🩸 BIRT RNAseq | 🧠 MRI | 🧪 Autoantibody | |
|---|---|---|---|
| Price | $300 ($16 COGS) | $1,600–8,400 | $730–6,200 |
| Data / test | 63K+ features | 1 image | 1 analysis |
| MS vs NMO | 91.3% AUC | Subjective pattern reading | Misses 25% seronegative |
| MS Subtyping | 78.8% AUC | Retrospective only (2–5 yrs) | No test exists |
| Early MS Screen | 75.4% AUC · NPV >98% | Gold standard but $3K–8K | No test exists |
| CNS inflammation | ✗ Blood can't see CNS | ✓ Direct imaging | No |
| Peripheral immune | ✓ Leading indicator | ✗ Lagging (structural) | Partial |



